ժҪ����ú���Ǿ���һ�����Եĺ���6%̿����״ϸС��������ˮ�����ʾߵ������������������÷�ú�Ҷ���ҵ��ˮ���д�����ν�Է��ηϣ��Ҵ�����ˮ���õ�Ч���á����ѵõ��йؿƼ���Ĺ㷺���ӣ������÷�ú�ҶԷ�ˮ�������������˴������о�������ȡ���˿�ϲ�ijɼ����о��˷�ú������м������ش��������ˮ�ķ�����̽�������û������������ʵ�鿼�����ڶ�̬�����С����pHֵ��ú���Ⱥ�ˮ��ͣ��ʱ�������ص�Ӱ�죬���ҶԷ�Ӧʱ���ú�������˵�����Ӱ��ʵ�顣�Է�ˮ�Ľ�ˮŨ�Ⱥͻ���������ʵ���о���ʵ������������pHֵΪ8.00��ú����Ϊ1��ˮ��ͣ��ʱ��Ϊ30min����ˮŨ��Ϊ15mg/l�ͻ�����Ϊ5��1ʱ����Ч����ã��ɴ�807%���ŷ�Ũ�ȿ��Կ�����2mg/l���ڣ��ﵽ���������ŷű���
����
ˮ�����������ܽ��Ե�H2S��HS-��S 2-���ʹ������������еĿ��������������Խ��������Լ�δ�����л�����������������״�ˮ��ɢ���ڿ�����������ζ���Ҷ��Ժܴ���������������ϸ��ɫ�ء�����ø�����������еĶ�������ã�Ӱ��ϸ���������̣����ϸ����֯ȱ����Σ���˵�����������������ܸ�ʴ�����⣬�����Ա���ˮ�е��������������ᣬ������ʴ��ˮ�ܵ��ȡ���ˣ�������ˮ����Ⱦ��һ����Ҫָ�ꡣ
�ڹ�ҵ�����������κ�����¶���������������ŷ����⡣ʹ����������Ĺ����������ܷ��ϵͳ�л��Ч�ŷ���н��С��������纬��0.1%��H2S�ͻ�Ѹ������ͷʹ����ѣ�������������H2S�ͻ���������������������Ӵ������������ж�������о��仵�����ݡ�ͷʹ�ȡ���ҵ�ϣ������в���H2S�������ó���0.01mg/L��
��1��ʵ�鲿��
1ʵ������
PVC���ӽ����������ӽ�������ˮ��2̨����ֽ����ϴ������Ӳ��������25ml��50ml��ʽ�ζ��ܡ���ƿ����Һ�ܡ�����ƿ����ֽ����ϴ������Ӳ��������������ϸ�����������ҷ���ֽ���ζ��ܼܣ�4�ס�150ml��250ml����ƿ��25ml��50ml��ɫ�ζ��ܡ�PHS-3C����pH�ǡ�
�Լ�������
�����=19g/ml�������=69g/ml��������Һ�������������ơ���������Һ��������������Һ��C=1mol/L����20g������������250mlˮ�У���ȴ�����£�ϡ����500ml���ظ���ر���Һ��C=0.1000mol/L����ȡ105����2h�Ļ����ż��ظ����9030g����ˮ�У�ϡ����1000ml���ظ���ر���Һ��C=0.2500mol/L��ȡ105����2h�Ļ����ż��ظ����1258g����ˮ�У�ϡ����1000ml���⻯�ء����������C=0.1mol/L��
���ƣ���ȡ24682g��ˮ�����������C��0.2003g��ˮ̼��������ˮ�У�ת�Ƶ�1000ml����ƿ�У�ϡ�͵����̶ȣ�ҡ�ȣ�������ɫƿ�ڡ�
�궨����250ml����ƿ�ڣ�����1g�⻯�ؼ�50mlˮ������100ml�����ظ���ر���Һ��������Һ5ml������ҡ�ȡ���������5min���ô��궨�������������Һ��������Һ�ʵ���ɫʱ������1ml����ָʾ���������ζ�����ɫ�պ���ʧ����¼����Һ������ͬʱ���հζ���
���������Ũ��C(mol/L)����ʽ�����
C=0.1000��15.00��(V1-V2)
ʽ�У�V1�����ζ��ظ���ر���Һʱ��������Ʊ���Һ������ml����
V2�����ζ��հ���Һ����������Ʊ���Һ������ml����
0.1000�����ظ���ر���Һ��Ũ��(mol/L)��
��������Ʊ��ζ�ҺC(Na2S2O3)=0.1mol/L����ȡ10.00ml�����ձ궨���ĵ��������������Ʊ���Һ��100ml��ɫ����ƿ�У���ˮϡ�͵��̶ȣ�ҡ�ȣ�ʹ��ʱ���ơ�
�����ҺC(1/2I2)=0.1mol/L����ȡ12.70g ����500ml�ձ��У�����40g�⻯�أ�������ˮ�ܽ��ת����1000ml��ɫ�ܽ�ƿ�У�ϡ�������ߣ�ҡ����������ɫƿ�б�����á�
�����ҺC(1/2I2)=0.01 mol/L����ȡ10.00 ml�����Һ��100ml��ɫ����ƿ�С���ˮϡ�������ߣ�ҡ����ʹ��ǰ���ơ�
��ú��
1g�����Ե��ۣ��÷�ˮ�ܽ⣬�ڶ��ݵ�100ml,��ȴ���á�
�� 1‑1��ú�ҵĻҳɷַ���/%
|
ú�ҵ�Ԥ�����ô���������Һ����24h��
15����м
��Դ���ߺ��л�����������
Ԥ������ÿ��ʹ��ǰȡ�����ĸ����ۼ���������ϴ�·�ϴ�����Σ���ÿ�γ�ֽ�����ϴ��û�к�ɫҺ��Ϊֹ����ʹ��1+3������ϴ����û�л��ǵ�Һ��Ϊֹ����ʹ������ˮƯϴ���α�֤ȥ�����۱���û�����������
1.3ʵ���������
1.3.1����IJ���
����Ҫ�����ĸ�����������10.00ml0.01mol��L�����Һ���ټ�5ml������Һ���������ȡ��ڰ�������10min����0.01mol��L��������Ʊ���Һ�ζ�����Һ�ʵ���ɫʱ������1ml����ָʾҺ�������ζ�����ɫ�պ���ʧΪֹ��
1.3.2�����ʾ
�������ﺬ��ci(mg��L)����ʽ���㣺
|
|
ʽ�У�V0�����հ������У���������Ʊ���Һ������ml��
Vi�����ζ����ﺬ��ʱ����������Ʊ���Һ������ml��
V�������������50ml��
16.03����������(1/2S2-)Ħ��������g/mol��
c������������Ʊ���ҺŨ�ȣ�mol��L����
1.3.3�ŷű�
��ʵ���������Ķ�����S2-,���ʼŨ��ԼΪ10mg/L�����ŷ�Ũ��Ϊ�����������������������ŷű���2mg/L��
1.4�հ�����
��˫������ˮ����������ȡ50ml������ⶨʱ��ͬ������Լ�������1.3�� ����������пհ����顣
�궨Na2S2O3�ǵ����������15.7ml
��C Na2S2O3=0.1��15��(15.7-3)
=0.118mol /L
��������Լ���Na2S2O3�ı���Һ��Ũ����0.00118 mol /L
�ô�Ũ�ȵ�
�k=C��16.03��1000��V
=3.8468
k���ڴ�ʱ�����ͨ��ֵΪ3.8468
��2�� ʵ��������������
2.1����������ʵ�飺
��ʵ��Է�ˮpHֵ����ú�Һ���м�ıȺͷ�Ӧʱ�䣬����������ˮƽ����ʵ�飬ˮƽ��������ʵ�������2-1�ͱ�2-2��ʾ��
�� 2‑1��̬����ʵ������ˮƽ��
| |||||||||||||||||||
�ֲ�ʵ�鲽�裺
1.ȡ����Na2S��Լ1mol/l����Һ2mL���ݵ�2000mL��������pH������Ҫ����ֵ��
2. ȡ��Լ1000mL����ķ�ú�ң������ɵ�Һ�����24h�����á�
3. ȡԼ��400mL�������м������������ķ�������Ԥ������
4. ����ʵ���Ҫ���ʪ�ķ�ú�Һ�ʪ����м���������ȣ���������ֵĽ���ʹ֮���Ȼ�ϡ�
��̬����ʵ�����ݼ�����
�� 2‑2 ��̬ʵ����������L9������������
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
��ֱ�۷�����������ʵ�������з�����XiΪij���ص�iˮƽ��ʵ��֮�͡����ݱ�����A��B��C�����ص�Xiֵ���������Ximax��Ximin���������˵�������ض�ȥ����Ӱ����ɴ˵ó�Ӱ���С˳��ΪC>B > A,��ˮƽ���ΪA2�� B3��C2������ˮ�ý�ˮpHΪ8��ú����Ϊ1��2��ˮ��ͣ��ʱ��Ϊ30min��
2.2 ������ʵ��
����ʵ�鼫�������֪������ˮƽ���ΪA2�� B3��C2����Ӧʱ�䳤����м�����࣬S2-��ȥ���ʾ�Խ�ߡ�Ϊ����֤��Ӧʱ�����м��������Ӱ�죬���������ؽ����˵�����Ӱ��ʵ�顣
2.2.1 ˮ��ͣ��ʱ��Դ���Ч�ʵ�Ӱ��
����ΪpH=8.00��ú����=1��2������£��ֱ�����20min��30min��40min����ʵ��ʵ��Ӱ�����ء�
�궨Na2S2O3���������Ϊ��8.4ml----24.2ml
��ʵ����Եó�ʱ��̴���Ч�������п�����һ����Ӵ�ʱ��̫�̣���һ�����ǿ�ʼ�ķ�ˮ�����Խϲ����¡���ʱ�䵽30min��ȥ������51.64����Ϊ48.41����������Ϊ3.23������������Ϊ�������䣬��ˣ���ˮ�����ͣ��ʱ��Ϊ30min��
��ͬŨ�ȶԴ���Ч����Ӱ��
��ȷ����pHֵ����Ӧʱ�����м�ȣ�����Ч�ʻ��ܵ�����ǰ��Ũ�ȵ�Ӱ�죬ȡ�Ѿ����Ƶ�������Һ��2.1.3.2.9��2ml��2.3ml��2.6ml��3.5ml��
����������ˮ��ͣ��ʱ�䣺30min
pHֵ��8.00
ú���ȣ� 1��2
�� 2‑5 ��ͬŨ�ȶԴ���Ч����Ӱ��
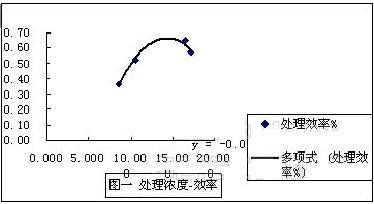
ͼ 2‑1 Ũ�Ⱥʹ���Ч�ʵĹ�ϵ
|
��ͼ���Եó���Ũ�ȱ仯ʱ����Ч��Ҳ��Ӧ�ı䶯����ͼ�����Եó����̣�y=-0.0091��X2+0.261X-1.2062 �ɴ˿��Ի���֪����ѵ�Ũ����14.5mg/l����ʱ�Ĵ���Ч�������66.5%��
�ڸ�Ũ�������ڷ�Ӧ�ĽӴ�����ͽ���ʱ�����ޣ����ܳ�ֵķ�Ӧ��ֱ�������������һ����Һ�Ķ�·������δ��Ӧ��ֻ�в��ַ�Ӧ�ĵ�Һ��ֱ���ŷţ�ʹ���ŷŵ�Һ��Ũ�����ߣ�����Ч��Ҳ���½��ˡ�
2.2.4 ȷ����ѵĻ�����
�ڹ����ϲ������������������������ǿ���ʽ�����ǻ���ʽ�ģ�����������䴦��Ч��
2.2.4.1 ʵ������ ˮ��ͣ��ʱ�䣺30min
pHֵ�� 8.00
ú���ȣ� 1��2
Ũ�ȣ� 14.5mg/L���������ֵ�Ա궨Ϊ��
ʵ��ԭʼ���ݼ����ݴ���
��������ͬ��������������ʵ��
�� 2‑6 ȷ����ѵĻ�����
�������ϵļ������ͱ����Եó�ͼ��
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
��ˮŨ����13.4mg/l������Ч����65�����㣬��ˮŨ����1.5mg/l���㣬�����ۻ������ǣ�2.3�����ɱ���ͼ�ó�ʵ�ʵ���ѻ�������5��1����ԭ����Ҫ����Ϊ�ڷ�Ӧ�Ĺ�������δ��Ӧ�ַ�Ӧ����Һֱ�������˳��ڡ�����ѵ�Ũ�ȡ� pHֵ��ú���Ⱥ�ˮ��ͣ��ʱ��������·�Ӧ������ֵ��ʵ��ֵ֮����һ����
2.3 ��֤��ʵ��
ʵ��������pHֵΪ8.00��
ˮ��ͣ��ʱ��Ϊ30min��
ú������1��2��
��ˮŨ����14mg /l��
��������5��1��
����Ҫ�����ĸ�����������10.00ml0.01mol��L�����Һ��2.1.3.2.3�����ټ�5ml������Һ(2.1.3.2.12)���������ȡ��ڰ�������10min����0.01mol��L��������Ʊ���Һ��2.1.3.2.10���ζ�����Һ�ʵ���ɫʱ������1ml����ָʾҺ��2.1.3.2.15���������ζ�����ɫ�պ���ʧΪֹ��
�궨Na2S2O3��Ũ�������ĵ����Ϊ��16.4ml����31.5ml �� ��V=15.1ml
��C Na2S2O3=0.1��15��(15.1-3)
=0.124mol/L
��������Լ���Na2S2O3�ı���Һ��Ũ����0.0124 mol /L
�k=
k���ڴ�ʱ�����ͨ��ֵΪ3.983
Ũ�ȵĹ�ʽ��Ϊ C= k��(8.8��0.0126��0.0118-V) V������Na2S2O3���
���Ѿ�������ѵ�Ũ�ȡ� �����ȡ� pHֵ��ú���Ⱥ�ˮ��ͣ��ʱ��������£����ˮ�ŷ�Ũ�ȿ��Կ�����2 mol/L���ڣ�����������ŷű���
��3�� �������⼰չ��
3.1 ��������
�������������Կ�����ú�����������ˮ����⼼���ǿ��еġ���Ϊ���Է��ηϵ��ص㣬�ڻ����������ž�Ŀ���DZ�ܺ�����Ӧ��ǰ��������ʵ��Ӧ�÷����д����ķ�������⣺һ�Ƿ�ú�ҵĴ�Ч����Ҫ��һ����ߣ����ǻ�ˮ�������⣻�����ڹ����ϼ���ļ�����������ȷ�������������о���������,�Է�ú�ҵ�����������ѧ���о�������������������о���ͣ����ʵ���ҽΡ�
3.2 չ��
������ʵ��Ӧ�÷�����ڵ��ķ�������⣬Ҳ���Ƿ�ú�������Ҫ�����о����ĸ�����
3.2.1 ��һ����߷�ú�ҵĵ��Ч��
��߷�ú�ҵĵ��Ч�ʣ�����ѡ��һЩ��̿���ϸߵķ�ú�ҡ�������Ҳ������150Ŀ���ڣ�����̫����Ƿ�ú���е��������ɷֺ������IJ����е�����õ����ʺ����Ӵ������ڷ�Ӧ�Ĺ����з�ú��û����ʧ�������ڷ�ˮ����������߲��ֵķ�ú�ҡ����Ҳ��öԷ�Ӧ�ķ�ú�ҵ���ҲҪ���������IJ��䣬�Ա�֤��ӳ�ڿ��Ƶķ�Χ֮�ڡ�
3.2.2 ���ˮ����Ч��
Ŀǰ�ڹ����ϳ���������������ʵ�ֻ�ˮ�����⣬����һЩ������ˮ���뷨Ҫô�ɱ�����Ҫôʵ����һ�����ѡ��ڱ�ʵ���о��������������������Ƕ�ʵ������һ����Ӱ�졣����ij�̶ֳ���Ҳ�谭�˸�������ƹ�Ӧ�á�����Ѱ��һ�ֱȽϺõĻ�ˮ���뷽���Ƿdz���Ҫ�ġ�
3.2.3 �����ϼ���ļ�����������ȷ��
���ڣ���Ӧ�IJ��Ͻ��У���Ӧ���е���м����Խ��Խ�٣��������1��2���ң���ƫ���ֵ�÷�Ӧ�ж����в�����Ӱ�졣��ʵ��ȷ�������ߵ������Ϊ1��2�Ǻ����ѵģ�ֻ���ڳ�ʱ��Ĺ���ʵ�ʲ����еIJ��ϵĻ��۲ſ�����һ����ŵ���ֵ��
3.2.4 ��ǿ�����о�
�����о���������,�Է�ú��������������ѧ���о���������,�����о���ͣ����ʵ���ҽΡ�����Ҫ���ӷ�ú��������ˮ�Ĺ��̻�������Ӧ����ѧ�����۵��о���ֻ���������ܹ�����߷�ú�ҵ�ú��Ч�ʷ�������ͻ�ơ����⣬Ӧ��ǿʵ���Լ����������豸���о��뿪����ʹ��ѧ��������ת��Ϊ���������Ӷ��ٽ���ú���ڷ�ˮ����������ƹ���Ӧ�á�